Scientists have grown dementia in a dish
But we wouldn't recommend eating it; you'd ruin the experiment, not to mention your appetite
IF revenge is a dish best served cold, then consider dementia: the degenerative brain disease has surely more than earned a little pay back.
Unfortunately, applying such anthropomorphism to dementia is just plain silly; as is suggesting getting our own back on the condition, but studying its foibles to maybe some day nip it in the bud is quite another matter.
Ha! Take that, dementia.
That's why scientists are growing dementia in a dish; more accurately, growing 'mini brains' or organoids in a petri dish. It's all to do with understanding the impact of genes; one in particular, that is linked to some forms of dementia and other age-related diseases.
The gene in question is called Angiogenin or ANG, and it is associated with a number of neurodegenerative diseases commonly associated with our elder years. These include fronto-temporal dementia (FTD), motor neurone disease (MND), and Parkinson's Disease.
But sadly not vascular dementia – another common form. Still, you can't have everything. Besides, for that one, scientists would have to grow a vascular system, too. Probably a step too far; stick to the organoid 'brains'.
Brain in a bottle
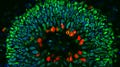
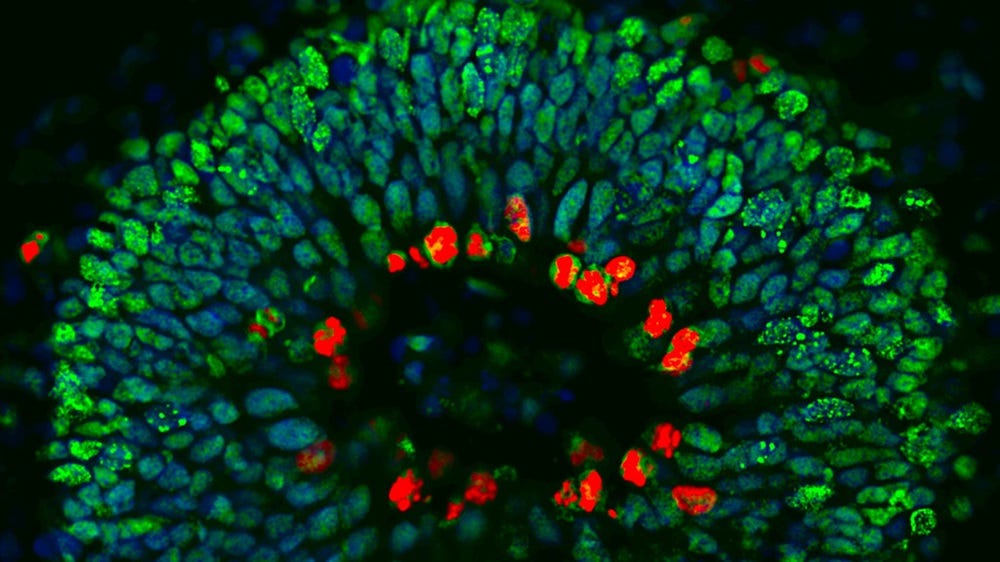
A mini-brain is a tiny 3D structure grown from clusters of human stem cells. It provides scientists with a realistic model to study the step-by-step development of disease. It also provides an ideal structure on which to screen drugs. In this case, it also proved an ideal method to test the impact of dodgy genes.
In its healthy state, as the scientists at the University of Bath have found, ANG plays an important role in the pace at which undifferentiated stem cells develop into specialised nerve cells.
In its mutated form, however, ANG causes stem cells to persist in their original state longer than they should. In lab experiments, this slowing down of the differentiation process was seen to result in striking defects in the nerve cells once they had reached their adult form.
“This suggests nerve-cell degeneration may be primed by defects occurring during early development,” said Dr Vasanta Subramanian, from Bath's Department of Life Sciences, who led the research.
In earlier work, the same research group found that ANG, in its healthy form, protects nerve cells against damage, degeneration and impairment of function.
By contrast, the mutated form of the gene causes nerve cells to be more susceptible to stress (a natural occurrence as cells age and experience wear and tear), leading to premature cell death.
“Our new discovery adds to our understanding of Angiogenin and its importance in protecting us from diseases associated with ageing,” she added.
For their latest work, the researchers studied a family affected by both fronto-temporal dementia and motor neurone disease. Genetic tests showed that some of the family members had mutations in ANG while others did not.
The scientists grew a 'mini brain' for each of the family members, using their individual stem cells. The researchers then observed striking neurodevelopmental defects in the mini-brains of family members carrying the ANG mutation.
“This seems to indicate that subtle development defects play a role in disease susceptibility or onset,” said Subramanian. “I envisage a time when we will be identifying people who are susceptible to these diseases, screening them for genetic mutations and offering early-intervention gene therapy to fix the defects.”
Subramanian said more research was needed to fathom mechanisms by which ANG acts to protect cells, and to better understand its function in stem cells.
Animals not included
Understanding –and some day treating – the role of faulty genes in the development of neurodegenerative conditions isn't the only purpose of the research. Aside from the general quest for knowledge, it also has a further laudable aim – to reduce the use of animal testing in research.
The work was funded by grants from the National Centre for the Replacement, Refinement & Reduction of Animals in Research (NC3Rs), BRACE, and the Wellcome Trust VIP award.
Chris Williams, chief executive of BRACE, said: "We applaud Dr Subramanian’s innovative research, which could make a big difference in tackling fronto-temporal dementia. Better understanding of the Angiogenin gene and its link to FTD could support treatment to slow down or stop the disease in the future.
“This type of dementia tends to have an early onset between the ages of 45-65 years, and often has a devastating impact during middle age. We are hopeful that this BRACE-funded research may play a key role in one day reducing the impact of the condition."
Dr Jessica Eddy, NC3Rs regional programme manager, added: “Research into the brain and neurological disorders relies in large part on animal models, and it is fantastic to see Vasanta’s 'mini-brain' organoids delivering new insights into neurodegenerative diseases.
“It is testament to the utility of these models that they are still being applied to new research questions, almost 15 years after we awarded Vasanta the initial funding to develop human cell-based alternatives to the use of animals in ALS (the most common form of motor neurone disease) research.”
The study was published in the Journal of Pathology.
Main image: A section through a human brain organoid showing stem cells that contain protective antibodies (stained green and red). The cells' nuclei are stained blue. Credit: Ross Ferguson and Vasanta Subramanian. Courtesy of Bath University
MC